Clinical-Grade
Lymphatic Restoration.
LymphRestore is a Class I medical device delivering multi-modal energy for clinical lymphatic restoration. Programmable protocols, integrated data capture, and modular treatment heads enable precision therapy across a growing range of indication areas.

Precision engineering for clinical lymphatic therapy.
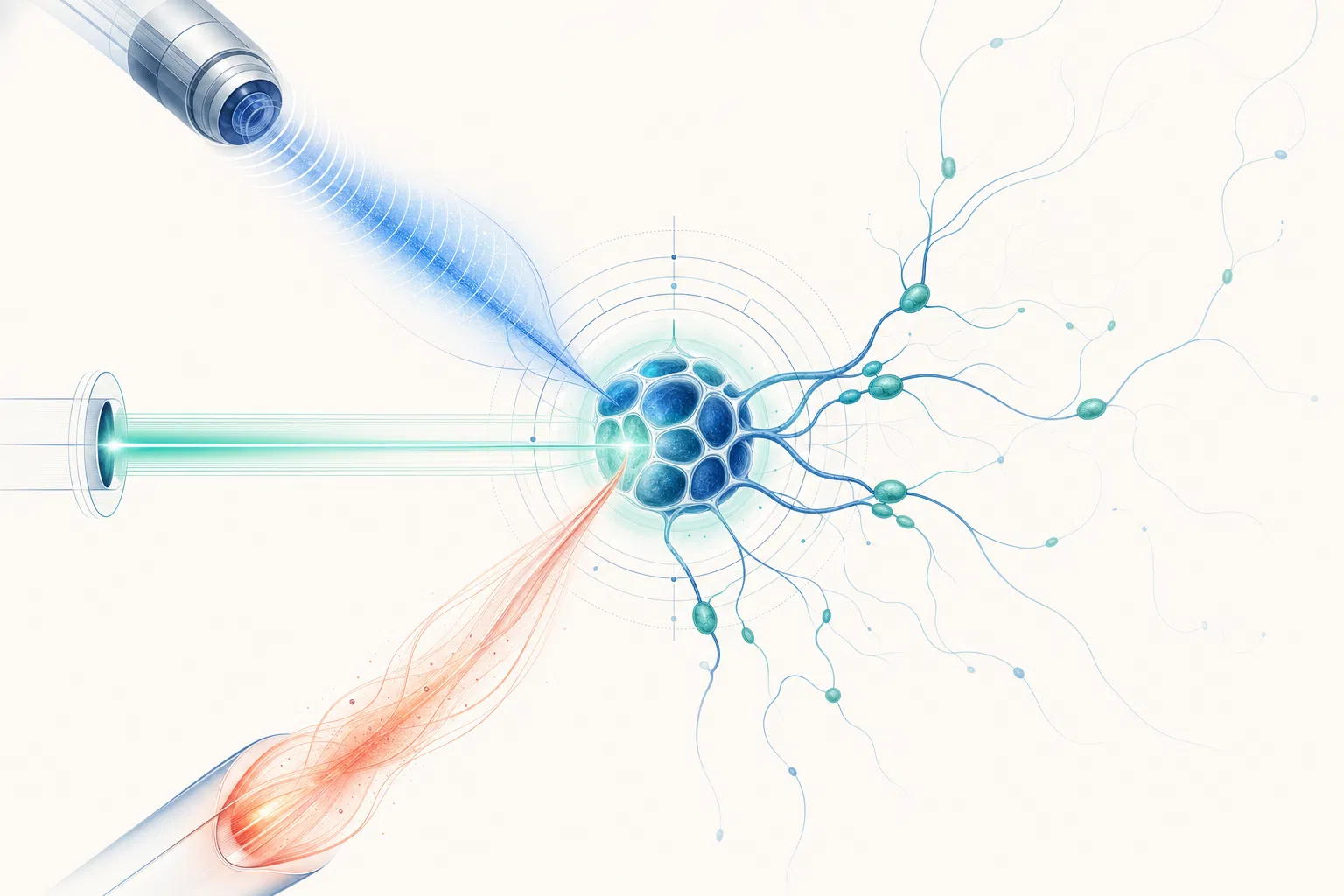
The LymphRestore system combines a programmable console with interchangeable treatment heads, delivering calibrated multi-modal energy across mechanical, photonic, thermal, and pneumatic modalities.
Four modalities. Programmable precision. Clinical evidence.
Multi-Modal Energy Delivery
Combines mechanical, photonic, thermal, and pneumatic energy modalities in programmable sequences for comprehensive lymphatic restoration.
Programmable Treatment Protocols
Clinician-defined treatment sequences with adjustable parameters for intensity, duration, frequency, and modality combination per session.
Clinical Data Capture
Integrated data logging captures treatment parameters, patient response metrics, and outcome measurements for longitudinal analysis.
Clinical-Grade Precision
Engineered to Class I medical device standards with calibrated energy delivery, safety interlocks, and real-time monitoring systems.
Research-Ready Architecture
Built-in data export capabilities support clinical trial protocols, enabling systematic evidence generation across indication areas.
Modular Treatment Heads
Interchangeable applicator heads optimized for different anatomical regions and treatment depths, from superficial to deep lymphatic structures.
From assessment to outcome — a systematic approach.
Assessment
Clinician evaluates patient lymphatic status using LymphLens diagnostic data and clinical examination to determine treatment parameters.
Protocol Selection
Select from pre-programmed treatment protocols or create custom sequences based on indication area, severity, and patient response history.
Treatment Delivery
Multi-modal energy delivery with real-time monitoring. The system adjusts parameters dynamically based on tissue response feedback.
Data Capture & Analysis
Treatment data is logged automatically. Longitudinal analysis tracks patient progress and informs protocol optimization over time.
Expanding the clinical frontier of lymphatic therapy.
Lymphedema
Primary and secondary lymphedema management through targeted multi-modal lymphatic stimulation and drainage pathway restoration.
Post-Surgical Recovery
Accelerated lymphatic recovery following surgical procedures involving lymph node dissection, reconstruction, or disruption.
Chronic Venous Insufficiency
Supporting lymphatic-venous interaction in patients with chronic venous disease and associated edema conditions.
Neurological Applications
Emerging research into glymphatic system support for neurodegenerative conditions through peripheral lymphatic optimization.
Urological Health
Pelvic lymphatic restoration for conditions including chronic prostatitis, erectile dysfunction, and lower urinary tract symptoms.
Orthopedic Recovery
Post-operative and chronic inflammatory condition management through targeted lymphatic drainage in musculoskeletal regions.
The clinical tier of a unified platform.
LymphLens
AI-powered lymphatic imaging and assessment
Learn moreLymphRelief
Mechanical and light-based lymphatic stimulation
Learn moreLymphRestoreCurrent
Clinical-grade lymphatic restoration
Interested in clinical lymphatic restoration?
Request the LymphRestore clinical brief for detailed specifications, indication protocols, and partnership opportunities.